Theme: Novel Strategies and Innovations in Pharmaceutical sciences
Asia Bio 2019
Conference Series LLC Ltd invites all the participants from all over the world to attend “International Conference on Bio-Pharmaceuticals” during Feb 28-March 01, 2019 at Osaka, Japan which includes prompt keynote presentations, Oral talks, Poster presentations, and Exhibitions. Asia Bio 2019 offers a best platform with its well organized scientific program to the audience which includes interactive panel discussions, keynote lectures, plenary talks and poster sessions on the topics Bio-pharmaceuticals sciences, Pharmaceutical Sciences, Pharmaceutical Chemistry and Medicinal Chemistry, Biomarkers and Biosimilars in Pharmaceutical Development, Pharmacovigilance and Cosmetovigilance, Medicinal Chemistry and Drug Discovery, Industrial Pharmacy, Pharma Analytical Techniques and Instrumentation, Radiopharmaceuticals, Pharmaceutics and Novel Drug Delivery Systems and many more.. The conference invites delegates from Biopharma laboratories, Pharmacists, Academicians, Clinicians, Researchers, Healthcare professionals, students, business delegates and Young researchers across the globe providing a better podium, interconnecting the latest research, technological developments in the arena as well as therapeutic aspects.
Why to Attend:
With all the scientific people over the world focused on learning about advancements in Pharma community. This is a best globalized opportunity to reach the largest assemblage of participants. We anticipate participants, renowned speakers and eminent delegates across the globe attending the conference to share their valuable presentation and galvanize the scientific community.
It is a perfect platform for researchers, scientists and, delegates to share experience, foster collaborations across industry and academia, and evaluate emerging technologies across the globe.
Our services have always met with great achievement in Business Conferencing. World-renowned speakers, the most recent and advanced techniques, developments, and the newest updates are the prominent features of the conference.
Target Audience:
- Researchers from Pharmaceutical Companies, Pharmacy Associations and Societies
- Students, Professors, Researchers, and Faculty of Pharmaceutical Sciences & Biological Sciences from Universities and Medical Colleges
- Scholars from Pharmaceutical and Biological Science backgrounds
- Directors, CEO’s of Organizations
- Researchers
- Graduates and post graduates in pharmacy
- Business development professionals, Consultants and Pharma service providers
- Health professionals
- Marketing, Advertising and Promotion Agency Executives, Solution Providers
Track 1: Bio-Pharmaceutical Sciences
Biopharmaceutical Science includes study of the drug discovery and their development, along with their properties, both physical and chemical. It also deals with the biological effects of drugs like onset and duration of action along with their intensity in the body. Biopharmaceutics help in calculating the bioavailability and therapeutic activity of the drugs in the body.
Biopharmaceutical science involves the creation of medical drugs by artificially growing some or all of their components. It is the pluridisciplinar background on the discovery phase of the drug development process, at the advanced level. This field includes the study of molecular mechanisms of disease, potential therapeutic targets and therapeutic strategies. The Session discusses recent trends in pharmaceutical research, in which increasing numbers of drugs are coming from large biopharmaceutical molecules. Bio-Pharmaceutical Sciences session includes talks on diversified fields like Biopharmaceutics and drug disposition, Innovations in clinical development, Pharmaceutical technology, Drug discovery and design, Pharmaceutics and drug delivery, Routes of administration, Fundamental drug development and etc.
Track 2: Biomarkers and Biosimilars in Pharmaceutical Development:
The recent challenges and approach for biosimilars in the biopharmaceutical industry will be discussed. The Clinical studies and the biosimilars innovator in the Pharmaceutical products can be known by the Clinicians for the research in the future of next generation biosimilars. Another major topic in the pharmaceutical industry Biomarkers in which the recent Biomarker research, development, and technology, measurable parameters for customized therapies will be discussed. The biomarkers are distinguished into Disease-related biomarkers which give an indication of the probable effect of treatment on the patient and Drug-related biomarkers indicate whether a drug will be effective in a specific patient and how the patient’s body will process it. The recent advances and the future scope will be discussed in this global Pharma summit.
Track 2: Pharmaceutical Sciences
Pharmaceutics is the study of relationships between per formulation, pharmaceutical formulation, delivery, disposition and clinical response. The inherent instability nature of a new drug will alter its desired form into undesired form when presented in a suitable dosage form with the excipients upon storage. In early days this process was confined only for assessing few characteristics, but today this process is being considered as a formulation strategy and hence tremendous technological advancement has been achieved in this field which enables us to save time and money through planned management system and hence impacts. Use of glorious statistical software even based on artificial neural networking is made the task of pre-formulating and optimization process easier. With the increasing number of novel and specialized compounds being developed, a "one size fits all" approach to drug formulation and delivery is no longer optimal, necessitating the consideration of formulations unique to each drug.
Track 3: Pharma Analytical Techniques and Instrumentation
Pharma Analytical Techniques are mainly important for manufacturing a medicine. Validation process should be done in every technique. Analytical method development and validation, Analytical IQ, OQ, and PQ are to be known in the analysis of the pharmaceutical product. Qualitative & quantitative elemental analysis and the current trends in the gravimetric analysis will be included in the analytical techniques. There are many techniques like UPLC and HPLC techniques, Electroanalytical methods, and Surface characterization techniques for chemical & physical analysis. Pharmaceutical Expo discusses all the analytical techniques and instrumentation that are used for the manufacturing medicine.
Track 4: GMP, GCP and Quality control
Good Manufacturing Practice is that part of quality management which ensures that products are consistently produced and controlled according to the quality standards appropriate to their intended use and as required by the marketing authorization, clinical trial authorization or product specification. Current good manufacturing practices guidelines, Current regulations, and quality standards are included. The role of Quality Impact Assessment & effectiveness checks is an essential operation of the pharmaceutical industry. CGMPs provide for systems that assure proper design, monitoring, and control of manufacturing processes and facilities. Management of product storage, transportation and distribution are the major aspects of the GMP practices.
Track 5: Pharmaceutical Regulatory Affairs
The Regulatory affairs also have a very specific meaning within the healthcare pharmaceuticals industries. It includes Regulatory requirements for pharmaceuticals and healthcare products, Intellectual property law for generics and innovator rights. Regulatory requirements for medical devices, Regulatory issues, quality control and business development and Novel strategies for growth in regulatory affairs are to be discussed. Intellectual property rights are also included.
Track 6: Clinical and Dispensing Pharmacy
Clinical Pharmacy includes all the services performed by pharmacists practicing in hospitals, community pharmacies, nursing homes, home-based care services, clinics and any other setting where medicines are prescribed and used. Role of Clinical Pharmacist, Clinical Pharmacy and its key role in treatment can be discussed. Dispensing pharmacy includes the preparation, packaging, labeling, record keeping, and transfer of a prescription drug to a patient or an intermediary. Pharmacotherapy and Environmental health hazards are also discussed.
Track 7: Pharmaceutical Industry
The innovative Pharmaceutical industry in the United States produces chemically-derived drugs developed as a result of extensive R&D and Clinical trials in both humans and animals. The innovator relies on patents and other forms of intellectual property rights to justify the investment required to bring a product to market (the U.S. patent term is 20 years). Several large multinationals produce the majority of innovative pharmaceuticals globally, but they have come to rely more heavily on research performed by industry partners, including smaller, innovative manufacturers. Pricing of innovative pharmaceuticals is designed to recapture R&D expenditures.
Track 8: Industrial Pharmacy
Industrial pharmacy is a discipline which includes manufacturing, development, marketing and distribution of drug products including quality assurance of these activities. The reasons for the increase of large-scale manufacturing Economic – As the scale of manufacturing batches increases so, proportionally, does the cost of production decreases Accuracy – The larger the quantities of materials involved so, proportionally, is the accuracy of measurements increased.
Track 9: Pharmaceutical Nanotechnology
Pharmaceutical nanotechnology has provided more fine-tuned diagnosis and focused treatment of disease at a molecular level. Pharmaceutical nanotechnology is the most innovative and highly specialized field, which will revolutionize the pharmaceutical industry in near future. Pharmaceutical nanotechnology presents revolutionary opportunities to fight against many diseases. It helps in identifying the antigen associated with diseases such as cancer, diabetes mellitus, neurodegenerative diseases, as well as detecting the microorganisms and viruses associated with infections. It is expected that in the next 10 years the market will be flooded with nanotechnology devised medicine.
Size reduction is a fundamental unit operation having important applications in pharmacy. It helps in improving solubility and bioavailability, reducing toxicity, enhancing release and providing better formulation opportunities for drugs. In most of the cases, size reduction is limited to the micron size range, for example, various pharmaceutical dosage forms like powder, emulsion, suspension etc. Drugs in the nanometre size range improve performance in a variety of dosage forms. Major advantages of Nanosizing include (i) increased surface area, (ii) enhanced solubility, (iii) increased rate of dissolution, (iv) increased oral bioavailability, (v) more rapid onset of therapeutic action, (vi) small amount of dose required, (vii) decreased fed/fasted variability, and (viii) decreased patient-to-patient variability.
Track 10: Medicinal Chemistry and Drug Discovery
Medicinal chemistry and pharmaceutical chemistries are disciplines at the intersection of chemistry, especially synthetic organic chemistry, and pharmacology and various other biological specialties, where they are involved with design, chemical synthesis and development for the market of pharmaceutical agents, or bio-active molecules (drugs). Current developments in medicinal chemistry, Diagnostic and bioinorganic medicinal chemistry are to be discussed. It includes Green techniques for organic synthesis and medicinal chemistry. Drug discovery is the process through which potential new medicines are identified. It involves a wide range of scientific disciplines, including biology, chemistry, and pharmacology. It includes Advances in Drug discovery, recent advances in fluorinated pharmaceuticals based on natural products. Plant and microbially derived drugs in Drug discovery are also discussed.
Track 11: Pharmacognosy and Phytochemistry
Pharmacognosy and Phytochemistry will be discussed to impart knowledge of alternative systems of medicine as well as modern Pharmacognosy. The drugs from natural products, crude drugs, and plant products its availability and toxicological studies are discussed. Pharmacognosy practice in the industry and the documentation procedures for the herbal drugs are important to know according to the guidelines. Phytochemistry is the study of phytochemicals and the subfield of Botany and Chemistry. The Analytical Techniques and Constituents elements in the phytochemistry (Phyto constituents) are the major topic to be known in the field of phytochemistry. It would serve as a valuable resource of information to other botanical and healthcare science students, researchers and industrialists in the field of herbal medicine. So the delegates from the field of Pharmacognosy and Phytochemistry from all over the world can attend this Pharmaceutical Expo India to explore the Research.
Track 12: Pharmaceutics and Novel Drug Delivery Systems
The field of Pharmaceutics is the major discipline in the Pharmacy in which the pre-formulation studies and accelerated stability studies included. Novel drug delivery systems are the method by which a drug is delivered can have a significant effect on its efficacy. The major challenges and the recent advances in the targeted drug delivery systems make an important role in the Novel drug delivery systems. The Lipid and Vaccine drug delivery systems and Nanotechnology are also included in this topic.
Track 13: Pharmaceutical Packaging
Packaging is one of the biggest industry sectors in the world, worth several billion. Pharmaceutical packaging represents a meager percentage of this colossal market. The global healthcare industry has seen a shift in paradigm and is now skewed toward effective and meaningful packaging. Packaging was considered as an afterthought which was needed merely in the final stages of manufacturing for many pharmaceutical companies about a decade ago. But of late, pharmaceutical packaging has quickly become an essential part of the drug delivery system as well as a core element of the marketing mix, through which manufacturers can differentiate their products from those of their competitors. As drug manufacturers approach the 21st century, they face a number of challenges that packaging can help them meet. The demand for pharmaceutical packaging is increasing and will continue to increase as companies rely more on packaging and labeling as media to protect and promote their products, increase patient compliance, and meet new regulations.
Track 14: Pharmacovigilance and Cosmetovigilance
Pharmacovigilance is known as Drug Safety is the pharmacological science relating to the collection, detection, assessment, monitoring, and prevention of adverse effects with pharmaceutical products. Cosmetovigilance is the on-going and systematic monitoring of the safety of cosmetics in terms of human health. The Drug safety and efficacy methods, Detection of adverse effects and other drug-related problems and Pharmacoepidemology and drug safety comes under the Pharmacovigilance. Cosmetovigilance includes Safety monitoring of medicinal products, On-going and systematic monitoring of the safety of cosmetics and implementation of Cosmetovigilance system.
Track 15: Radiopharmaceuticals
Radiopharmaceuticals used as Diagnostic and therapeutic agents. The main group of these compounds is used as the radiotracers to diagnose the diseased tissues in a body. Radiopharmaceutical preparation is a medicinal product in a ready-to-use form suitable for human use that contains a radionuclide. The radionuclide is integral to the medicinal application of the preparation, making it appropriate for one or more diagnostic or therapeutic applications.
Track 16: Pharmacy Practice
Pharmacy practice is the discipline of pharmacy which involves developing the professional roles of pharmacists. Ethics in pharmacy is also very important for the any Pharma Practitioner weather they are from industry or from research institutes or Hospital Pharmacists. The scope, new dimensions and global standards of Pharmacy practice can also be included in Pharma Summit. The recent advances and the new drug applications and fundamental drug development can help the Pharmacy practice for the implementation of new drugs. Clinical pharmacy is a branch of pharmacy which deals with the patient care and medication and promotes health, wellness, and disease inhibition.
Track 17: Current Trends in Pharmacology
The recent studies and the current trends in the Pharmacology are discussed. It starts with Clinical pharmacology in which it is the science of drugs and their clinical use. Recent advances in neuro and psychopharmacology and the recent toxicological studies and its issues are discussed for the effective knowledge in the branch of Pharmacology. Pharmacogenetics has been defined as the study of variability in drug response due to heredity. Most recently pharmacogenomics has been introduced with the Pharmacogenetics. Current advances in the pharmacology and physiology of top channels play a major role in the Pharmaceutical Industry.
Track 18: Pharmaceutical Drug development
Drug expansion is the procedure of creating a new pharmaceutical drug to the global market once a lead compound has been found through the procedure of drug discovery. It includes pre-clinical research on microorganisms and animals, filing for regulatory statuses, such as via the United States Food and Drug Administration for an investigational new drug to initiate clinical trials on humans, and may include the step of obtaining regulatory approval with a new drug application to market the drug.
Analysis of global market trends, with data from 2012-2014, and projections of CAGRs through 2019. A look at the present state of the drug discovery market, existing platforms and products on the market, and clear analysis of the competitive environments including new, potential venues for novel technologies and approaches and descriptions of various products such as Bioanalytical instruments, high-throughput screening devices, informatics, and microarrays. Analysis of the main positive and negative factors in each sector of the market, potential future trends, and emerging technologies across different segments of the industry.
The Asia Pacific is the third largest pharmaceutical market in the world after North America and Europe. Major driver for the pharmaceutical industry in this region is generics, aided by factors such as the aging population and impending expiry of patented drugs. Asia-Pacific Biopharmaceuticals market was worth $36.57 billion in 2018 and estimated to be growing at a CAGR of 10.05%, to reach $59.03 billion by 2023. The market is expected to register a CAGR of 8.5% during the forecast period (2018 – 2023).
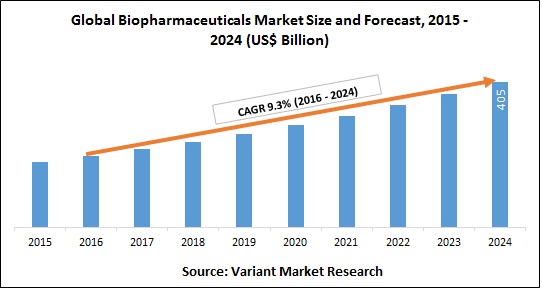
In 2009, Japan’s pharmaceutical market was worth $68.9 billion. It is projected to reach $72.0 billion in 2021, increasing at a Compound Annual Growth Rate (CAGR) of 0.3%. The medical device market was valued at $56 billion in 2016 and is projected to grow at a CAGR of 7.4% to $80 billion in 2021.
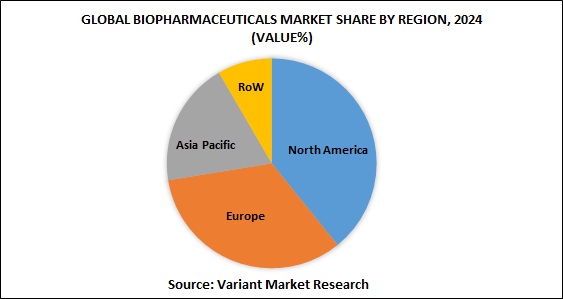
The Asia-Pacific biopharmaceuticals market is segmented on the basis of products into Monoclonal Antibodies (mAb), Erythropoietin, Biotech Vaccines, Recombinant Human (RH) Insulin, Granulocyte colony-stimulating factor (G-CSF), Interferon and Human growth hormones (HGH). Monoclonal antibodies segment holds the highest number of shares in the product sector. Based on therapeutics, the market is bifurcated into Neurology, Infectious diseases, Diabetes, Oncology, Cardiovascular and other therapeutic areas. In terms of therapeutic application, oncology is the largest segment in the biopharmaceuticals market.
Conference Highlights
- Bio-Pharmaceutical Sciences
- Pharmaceutical Sciences
- Pharmaceutical Nanotechnology
- Pharmaceutical Industry
- Pharmaceutical Packaging
- Radiopharmaceuticals
- Pharmaceutics and Novel Drug Delivery Systems
- Pharmaceutical Drug development
- Pharma Analytical Techniques and Instrumentation
- Current Trends in Pharmacology
- Biomarkers and Biosimilars in Pharmaceutical Development
- Pharmacognosy and Phytochemistry
- Pharmaceutical Regulatory Affairs
- Pharmacovigilance and Cosmetovigilance
- GMP, GCP and Quality control
- Pharmacy Practice
- Clinical and Dispensing Pharmacy
- Medicinal Chemistry and Drug Discovery
- Industrial Pharmacy
To share your views and research, please click here to register for the Conference.
To Collaborate Scientific Professionals around the World
| Conference Date | Feb 28-March 01, 2019 | ||
| Sponsors & Exhibitors |
|
||
| Speaker Opportunity Closed | Day 1 | ||
| Poster Opportunity Closed | Click Here to View | ||
Useful Links
Special Issues
All accepted abstracts will be published in respective Our International Journals.
- Pharmaceutica Analytica Acta
- Clinical Pharmacology & Biopharmaceutics
- Pharmaceutical Regulatory Affairs: Open Access
Abstracts will be provided with Digital Object Identifier by

















